Content
Fluid Properties
Vapor Pressure (Pv)
Fluid Properties
Vapor Pressure (Pv)
At a given pressure, a pure substance changes its phase at the saturation temperature, Tsat.
At a given temperature, a pure substance change its phase at the saturation pressure, Psat.
At a given temperature, the enclosed liquid in a container will evaporate. In phase equilibrium, the pressure exerted by its vapor is named vapor pressure, Pv. For a pure vapor, it is identical to the saturation pressure of the liquid at the given temperature.
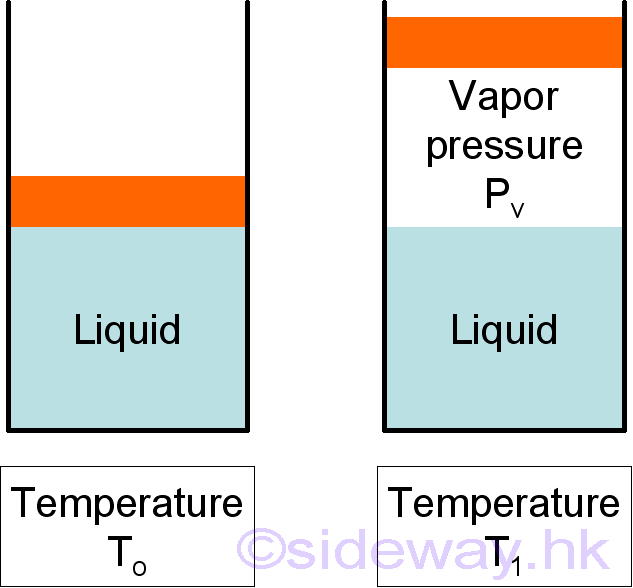
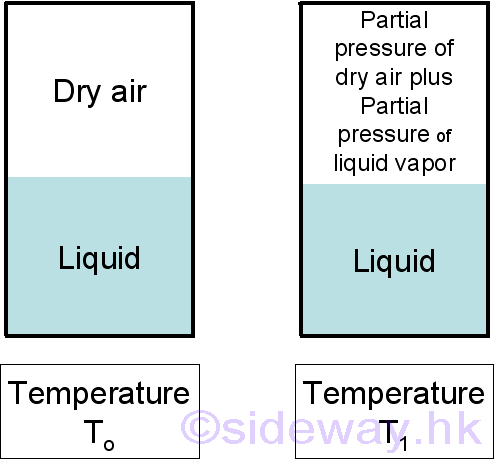
But when the vapor of the liquid is mixed with other gases, for example water vapor and dry air, the pressure caused by the water vaor is a partial pressure only. The atmospheric pressure will be the sum of the partial pressure of dry air and the partial pressure of water vapor.
In liquid flow system, the absolute liquid pressure may drop below the vapor pressure at some locations along the flow and cause unplanned locate vaporization, boiling and forms cavities in the liquid as vapor bubbles, named cavitation bubbles. As bubbles move away from the low-pressure regions, they will collapse and generate extremely high-pressure destructive waves. The formation and collapse of cavitation bubbles will cause the drop in performance and the erosion of component parts, for example irregular, narrowed flow passages of valve or pump, impeller blades of hydraulic turbine. And the formation and subsequent collapse of vapor bubbles in a flowing liquid is called cavitation.
©sideway
ID: 100100007 Last Updated: 2/3/2010 Revision: 0
Latest Updated Links
- Transportation International Airports Fukuoka FUK(last updated On 4/22/2026)
- Travel Singapore Sight Mandai(last updated On 4/16/2026)
- Travel Singapore Sight Sentosa Resorts World Sentosa(last updated On 4/16/2026)
- Transportation Bus(last updated On 4/12/2026)
- Travel Japan Prefectures and Climate(last updated On 4/8/2026)
- Travel Singapore Sight(last updated On 4/6/2026)
- Travel Singapore Sight Singapore Zoo(last updated On 4/6/2026)
- Travel Singapore Sight Rainforest Wild ASIA(last updated On 4/6/2026)
- Travel Singapore Sight River Wonders(last updated On 4/6/2026)
- Travel Singapore Sight Night Safari(last updated On 4/6/2026)
- Travel Singapore Sight Curiosity Cove(last updated On 4/6/2026)

 Nu Html Checker
Nu Html Checker  53
53  na
na  na
na
Home 5
Business
Management
HBR 3
Information
Recreation
Hobbies 9
Culture
Chinese 1097
English 339
Travel 51![]()
Reference 79
Hardware 55
Computer
Hardware 260
Software
Application 213
Digitization 37
Latex 52
Manim 205
KB 1
Numeric 19
Programming
Web 290![]()
Unicode 504
HTML 66![]()
CSS 65![]()
Selector 1
SVG 46
ASP.NET 270
OS 447![]()
MS Windows
DeskTop 7
Python 72
Knowledge
Mathematics
Formulas 8
Set 1
Logic 1
Algebra 84
Number Theory 207![]()
Trigonometry 31
Geometry 34
Calculus 67
Engineering
Tables 8
Mechanical
Rigid Bodies
Statics 92
Dynamics 37
Fluid 5
Control
Acoustics 19
Natural Sciences
Matter 1
Electric 27
Biology 1
